Base metal catalysts carrying PCP pincer ligands, where the central carbon is an sp2-hybridised carbene carbon atom, have found increasing attention in recent years due to their ability to perform difficult bond transformations. These ligand platforms enable these complexes to partake in metal-ligand cooperativity, which often endows them with reactivities comparable to their noble metal counterparts.
As part of a long-standing collaboration within this research area, the groups of Dr Young (National University of Singapore) and Dr Krämer (Maynooth University) have recently reported the results of a joint experimental and theoretical study in Angewandte Chemie International Edition.
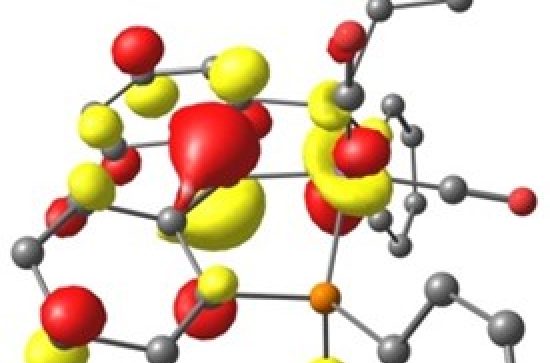
The Young group used a dehydration route to generate iron PCcarbeneP pincer complexes, which are normally synthetically difficult to access. These complexes are competent in activating H–H, aliphatic C–H and B–H bonds across their iron-carbene linkage. The PCcarbeneP pincer ligand was also found to stabilize multiple oxidation states, including unstable low-valent formally Fe(I) and Fe(–I) centres. Quantum chemical calculations were carried out by Dr Baptiste Leforestier, whilst previously on secondment to the Krämer group, to explore the reactivity and electronic structure of these iron pincer complexes. It could be shown that E–H bond activation is initiated via reversible carbonyl migration to the carbene position, representing an uncommon mechanism for metal-ligand cooperativity.
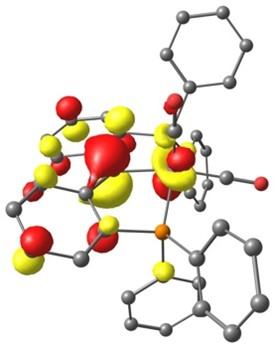
Singly occupied molecular orbital of [Fe–I(PCcarbeneP)(CO)2]–.
The ability of the PCcarbeneP architecture to stabilise a range of oxidation states at the iron centre in combination with its ability to partake in metal-ligand cooperativity demonstrates the utility of this base metal complex in catalytic applications.
The article is available from the following link:
“Access to and reactivity of Fe(0), Fe(–I), Fe(I) and Fe(II) PCcarbeneP pincer complexes”
https://onlinelibrary.wiley.com/doi/10.1002/anie.202104130
